A research team in the University of Macau’s (UM) Faculty of Health Sciences (FHS), led by FHS Associate Professor Zhao Qi, has developed a new nanozyme technology that can enhance the anti-tumour effects of chimeric antigen receptor (CAR) T cells by reshaping the tumour microenvironment. This technology can be used to improve the ability of the patient’s own immune system to fight the tumour. The study has been published in the high-impact academic journal Small.
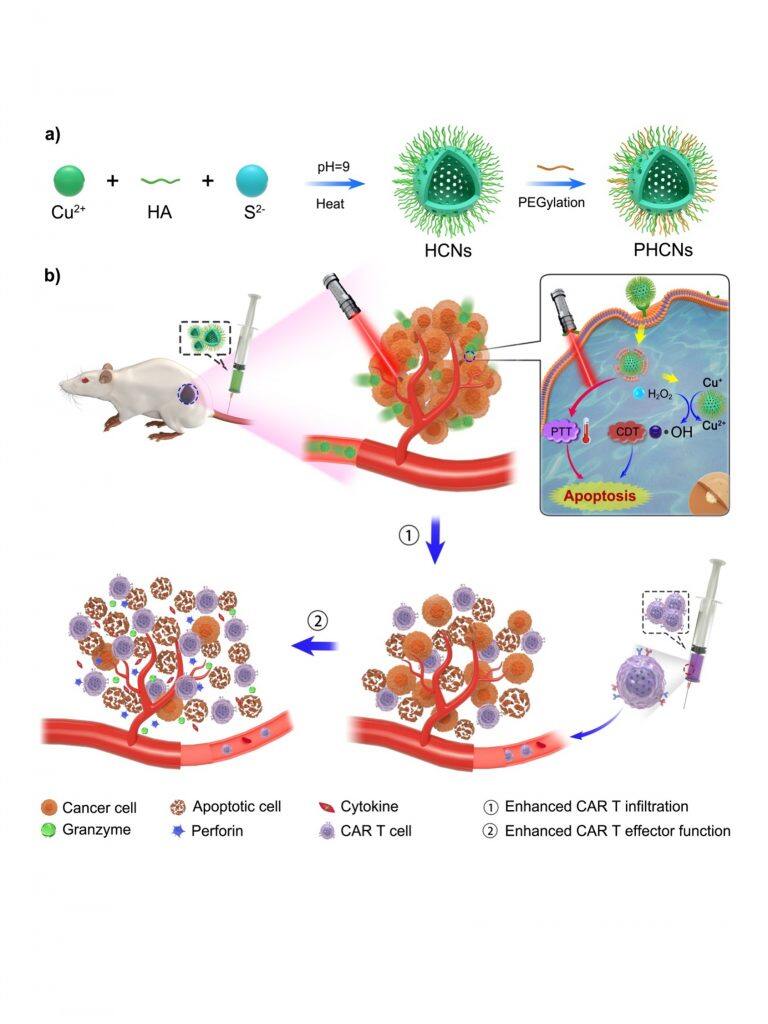

Adoptive cell therapy of engineered T cells with CARs represents an attractive and innovative strategy for cancer treatment. However, in solid tumours, one major obstacle limiting CAR T cell therapy is the tumour microenvironment (TME). Previous studies have reported that B7-H3 CAR cells based on non-small cell lung cancer (NSCLC) exhibited superior anti-tumour effects and inhibited tumour growth. However, B7-H3 CAR T cells partially eradicated solid tumours.


This study is a breakthrough in the treatment of solid tumours with CART cells. The team has found that one of the main obstacles limiting CAR T cell therapy is the tumour microenvironment (TME). Nanozymes exhibit merits modulating the immunosuppression of the tumour milieu. The team adopted a synergetic strategy by combining nanozymes and B7-H3 CAR T cells in solid tumours. This nanozyme with dual photothermal-nanocatalytic properties is endowed to remodel TME by destroying its compact structure. The team has discovered that the B7-H3 CAR T cells infused in mice engrafted with the NSCLC cells have superior anti-tumour activity after nanozyme ablation of the tumour. Moreover, the changes altered immune-hostile cancer environment, resulting in enhanced activation and infiltration of B7-H3 CAR T cells. This study clearly supports that the TME-immunomodulated nanozyme is a promising tool to improve the therapeutic obstacles of CAR T cells against solid tumours.
FHS’s PhD student Zhu Lipeng is the first author of the study, and Prof Zhao Qi is the corresponding author. FHS professors Liu Tze-Ming and Dai Yunlu made important contributions to this study. The study was funded by the Science and Technology Development Fund (FDCT), Macao SAR (file number: 131/2016/A3 and 0015/2018/A1), National Key R&D Programme of China (file number: 2019YFA0904400), and UM (file number: MYRG2019-00069-FHS). The full-text version of the related paper can be viewed at: https://onlinelibrary.wiley.com/doi/full/10.1002/smll.202102624
Source: Faculty of Health Sciences, University of Macau






































